Together for Integrated Response
Integrated Response Center
Medical Contact Centers face complex regulations, privacy laws, and a shortage of trained professionals. APCER Life Sciences addresses this issue by employing Healthcare Professionals (HCPs) who specialize in Medical Information (MI). They ensure a balanced and regulated response to inquiries from various stakeholders, including physicians, pharmacists, and patients.
Our global Integrated Response Center, with infrastructure in United States, Canada & India, equipped with state of the art technology platforms, provide end-to-end MI services based on customer requirements for a wide range of medicinal products. APCER’s unique and robust training program has made it a preferred choice for MI and contact center services. We ensure our professionals stay up to date on changing regulatory requirements through periodic training sessions. Contact Center operations can be scaled up and unexpected spikes in contact volume can be managed effectively with APCER’s MI function.
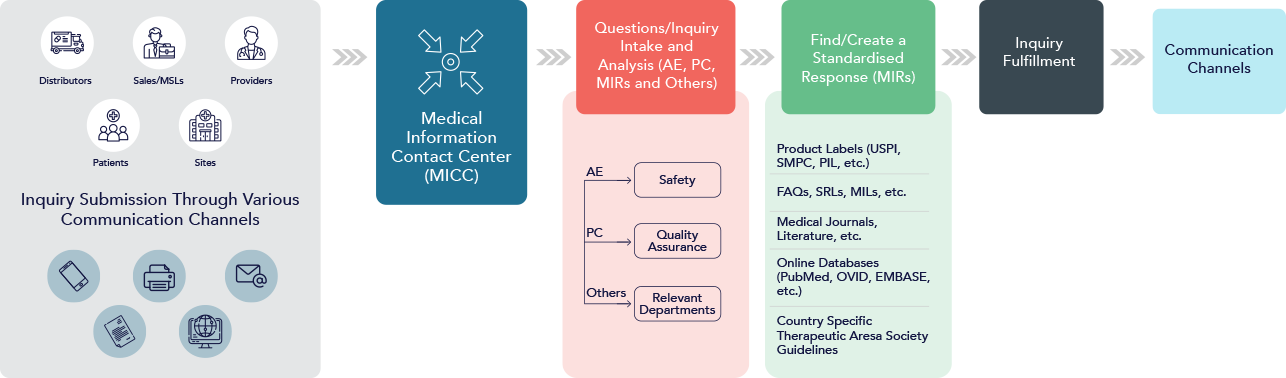
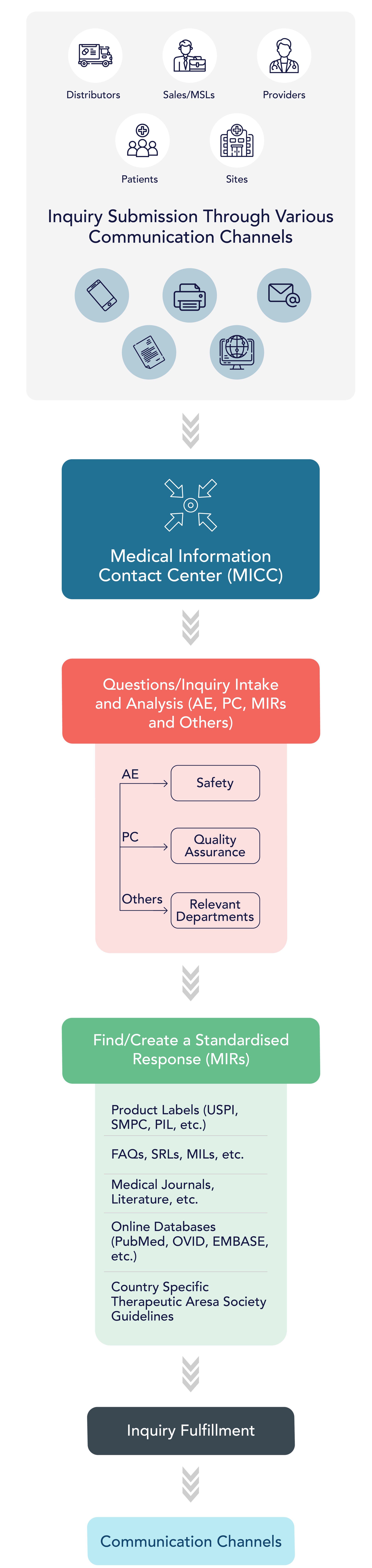
APCER’s Integrated Response Center receives, triages, and responds to information requests via telephone, fax, and email.
HCPs in our MI team, including physicians, pharmacists, and nurses, understand the nuances of a medical query and provide timely and accurate responses. APCER’s MI HCPs can communicate with the HCPs requesting medical information peer-to-peer. Patients and caregivers contacting APCER for medical queries are also assisted by the same HCPs. The requested information is presented in a way that can be easily understood by non-health care professionals.
The Integrated Response Center has a “Follow the Sun” approach to handling inquiries that keeps our team available even after normal business hours for all inquiries.
APCER’s MI team is cross-trained in pharmacovigilance procedures and relevant regulatory guidelines. We at APCER work closely with your pharmacovigilance team to collect, document, process, and report adverse events as a unit.
A trained expert ensures that the maximum amount of information is collected about an adverse event report at the initial point of contact. MI collaborates closely with our case processing team and performs follow-ups to ensure additional information is provided for effective adverse event reporting.
Reports of adverse events are documented in a fully validated MI database which has a dedicated Adverse Event module. Case processing teams can use this information to provide structured information to MI team members by capturing most of the information in dedicated fields.
The MI team at APCER integrates with its clients’ processes for handling product quality complaints (PQCs). The MI team members are trained on the most current guidelines for collecting and reporting PQCs.
As members of the APCER MI team, pharmacists have a deep understanding of industrial processes and can quickly assess the nature of the complaint and its probable cause. The members of the team collect as much information as possible from the complainant on the first contact to ensure that the complaint will be investigated effectively by the client. It is best to gather information at the time of the initial contact. APCER’s MI team members follow up with the complainants if additional information is required for further investigation.
Our MI team also assists in retrieving complaint samples by delivering prepaid mailers with return instructions to the complainants.
We have a fully validated MI database that includes a dedicated PQC module and contains all PQC-related information. APCER MI can work with its clients to configure the module for the best visualization of complaint data.
We develop and deliver standard responses, including FAQs and answers, and forward standard response letters as needed. APCER’s trained pool of HCPs can assist clients in creating quality MI letters for HCPs.
We can conduct literature searches to gather information about a subject. Our MI team works closely with its scientists and copy editors.
Content created can be managed within our fully validated MI database with its Content Management module.
MI team members often receive queries from HCPs that fall outside the scope of standard responses. Using literature surveillance and developing customized responses to HCPs’ medical inquiries, our MI function can assist physicians.
APCER hosts one of the world’s leading databases of medical information. The cloud-based system captures MI requests, adverse event reports, and PQCs through dedicated AE, PQC, QA, and Content Management modules. The system has been validated in compliance with 21 CFR Part 11 and EU Annex 11 and is hosted in a highly available and DR-enabled environment. In addition, our team has experience working with medical information systems hosted by our clients.
Our call center operations are managed by one of the leading call center solutions. The APCER Contact Center Solution is a cloud-based, next-generation omnichannel platform. In addition to HIPAA, PCI, SOX, and CPNI compliance, the solution is also compliant with many other security standards. It is fully validated as per 21 CFR Part 11 and EU Annex 11. It is hosted in a high availability environment (Four 9’s) and is equipped with DR capabilities.
Using both systems, our clients are able to generate standard reports/dashboards for the best possible data visualization.



