Together for
better health
Our Services

Pharmacovigilance
APCER offers complete, cost-effective, and agile solutions to challenges in clinical safety and pharmacovigilance. We tailor our resources to meet your budget and fit your culture.

Medical Information
APCER provides skilled healthcare professionals and a 24×7 Integrated Response Center to answer inquiries and disseminate information.

Regulatory Affairs
APCER helps clients develop a filing strategy and perform regulatory activities worldwide throughout the lifespan of a product.

Medical Writing
APCER’s team of medical writers helps life sciences companies craft medical, clinical and regulatory documents and stay in compliance throughout the product lifecycle.
Medical Affairs
APCER has the medical resources needed to listen to the market, respond to opportunities, and communicate with timeliness and accuracy.
Quality Assurance
APCER’s team of experienced quality auditors provide onsite and virtual GVP, GCP audits, GCLP audits and Gap Analysis services. Our end-to-end Quality Management System services ensure Anytime Inspection Readiness for our clients.
Suggested Content
Local Knowledge,
Global Compliance
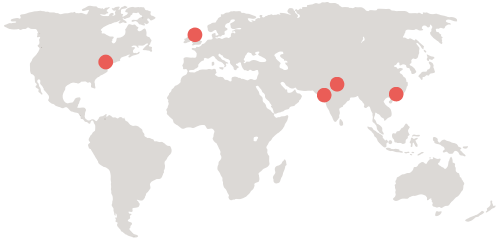
APCER Life Sciences, the truly global provider of safety, medical, regulatory, technology and quality assurance & auditing services. Our international team of physicians, pharmacists, scientists, communicators, and data specialists stand ready to respond to providers, patients, payers, and regulators, wherever you need cost-effective resources to collaborate with yours. We bring deep product knowledge, therapeutic experience, and best practices for worldwide compliance.
Each of APCER’s operational units in Asia, Europe, and North America serves as the regulatory intelligence and delivery center for its respective region. They continually track modifications in standards, guidelines, and regulations, incorporating their knowledge into standard operating procedures, working practices, and training modules that are shared globally.