Background
The Clinical Trial Regulation (CTR; EU No. 536/2014) and the Clinical Trial Information System (CTIS) came into application on January 31, 2022 in the European Union and European Economic Area. The CTR repealed the EU Clinical Trials Directive (EC) No. 2001/20/EC and national legislation that was put in place to implement the Directive. The CTR is adopted to overcome the identified gaps and to ensure a higher level of harmonisation regarding the authorisation of clinical trials in the EU.
The assessment and supervision processes for clinical trials throughout the EU are foreseen by the EU CTR via a centralised portal ‘CTIS.’ After the go live on January 31, 2022, clinical trial sponsors can register their trial in the CTIS, rather than in the European Union Drug Regulating Authorities Clinical Trials Database (EudraCT), although they may continue to use the EudraCT for a transition period. The CTIS will centralise the submission, assessing, authoring, and supervision processes for clinical trial application (CTA).
European Union Clinical Trial Regulation – What is new?
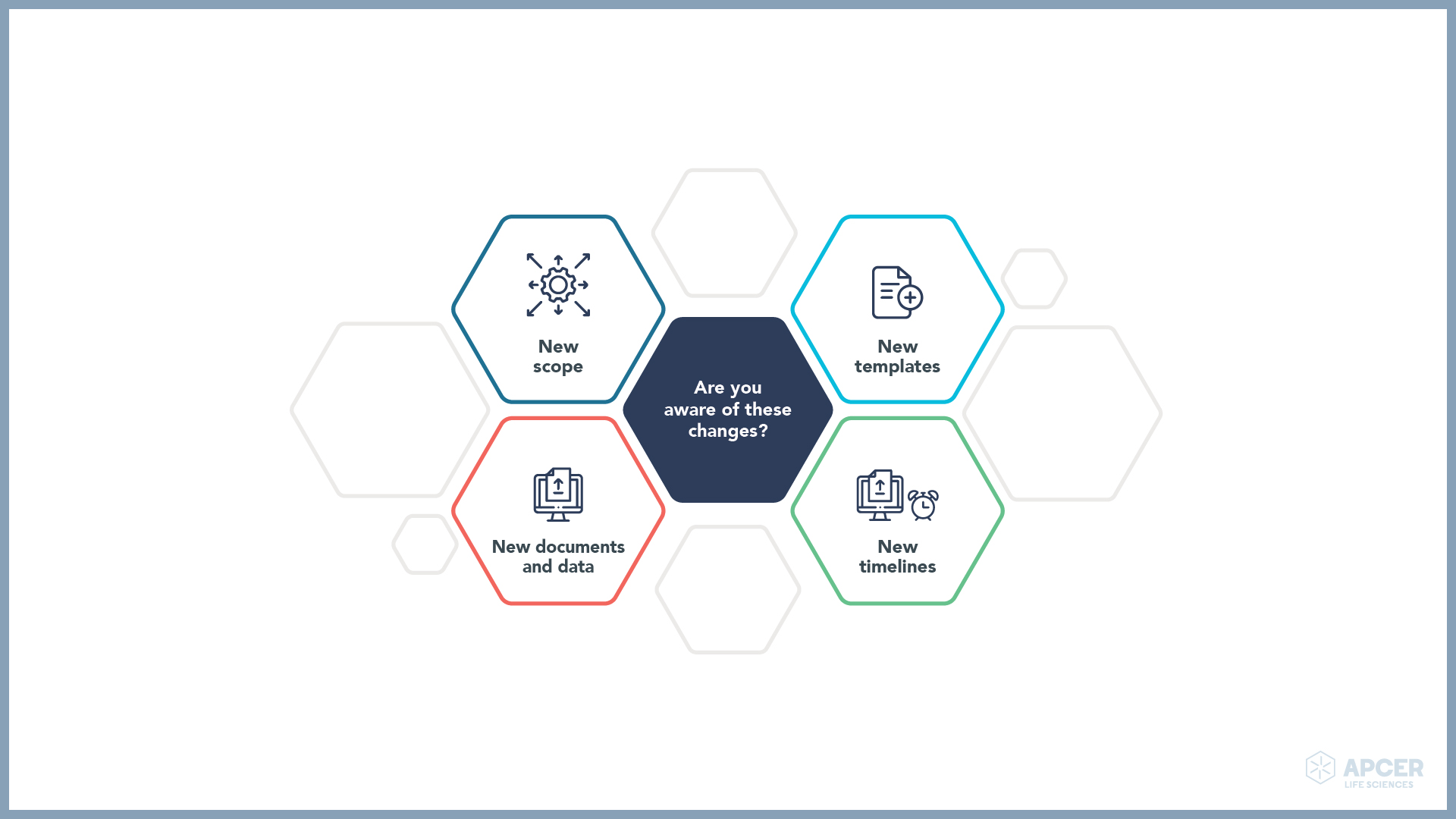
To harmonise the assessment and supervision of clinical trials and to make the process more efficient, the agency has expanded the scope and defined the way the data need to be submitted to the authority. Some of the key updates are highlighted below:
Are you aware of the transitional arrangements?
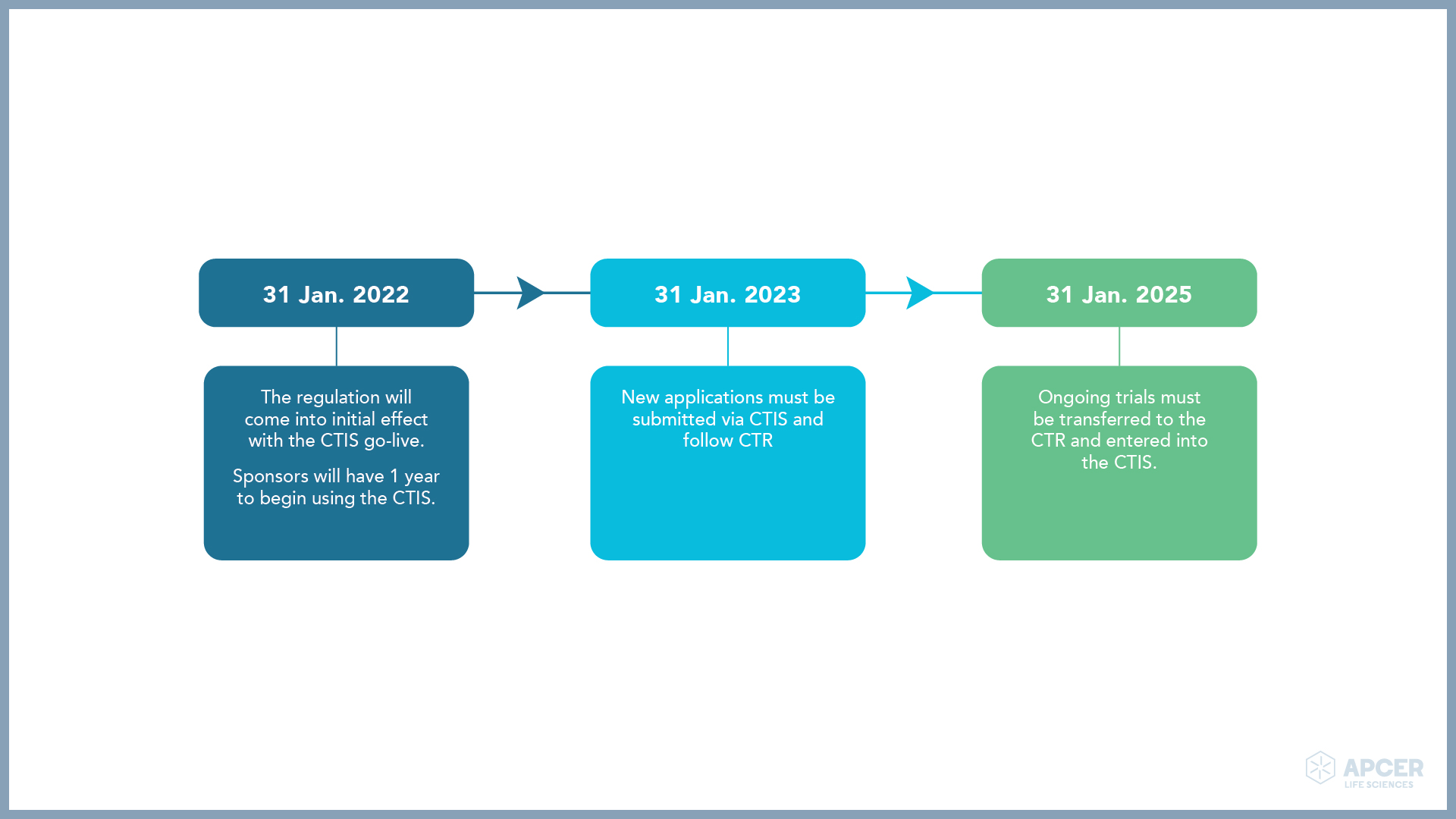
To facilitate the change from the directive (EU CTD) to the regulation (EU CTR), the regulation allows to start and conduct a clinical trial in accordance with the Directive 2001/20/EC (CTD) during a transitional period of 1 year after the CTR application date. Changes to the CTR have provided for a 3-year transition period, giving sponsors time to switch to the EU CTR.
Are you planning to conduct clinical trials or have ongoing trials in the EU?
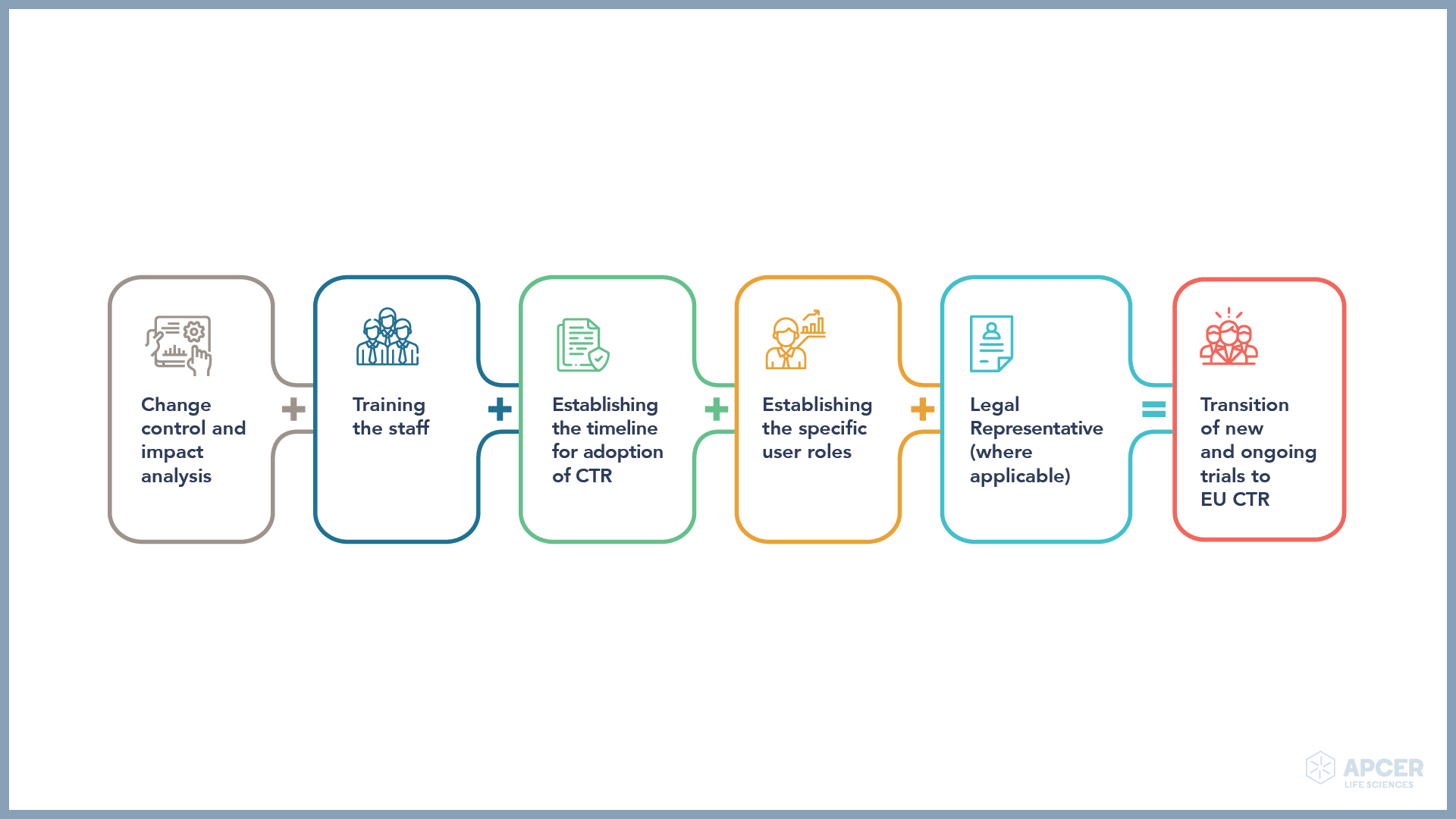
APCER understands that biopharma companies need to make necessary adaptions and take a step-wise approach to meet the regulatory requirements.
APCER’s EU CTR team can assist you in setting up the regulatory framework at every step to make the below-mentioned adaptations: