Overview
Pharmacovigilance (PV) at regional level has become an important topic of discussion in PV audits and inspections. For all European Union (EU) countries, relevant requirements are defined in Good Pharmacovigilance Practices (GVP) and applicable regulations. Besides these common EU requirements, there are also local requirements that need to be followed and met. Of the 30 member states in the European Economic Area (EEA), national competent authorities in 22 member states require the nomination of a PV contact person at regional level/local Qualified Person Responsible for Pharmacovigilance (QPPV) based on the authorisation/marketing status of a medicinal product. Similarly, countries like Australia, Canada, Russia, and the Kingdom of Saudi Arabia require the nomination of a ‘QPPV’ equivalent residing in the respective territory.
It is essential that a consistent and efficient decision-making system should be developed at regional level, which empowers the local QPPV in safety assessment in alignment with global PV functions. Global PV oversight of regional PV activities is needed to achieve consistent patient safety.
Challenges Encountered at Regional Level
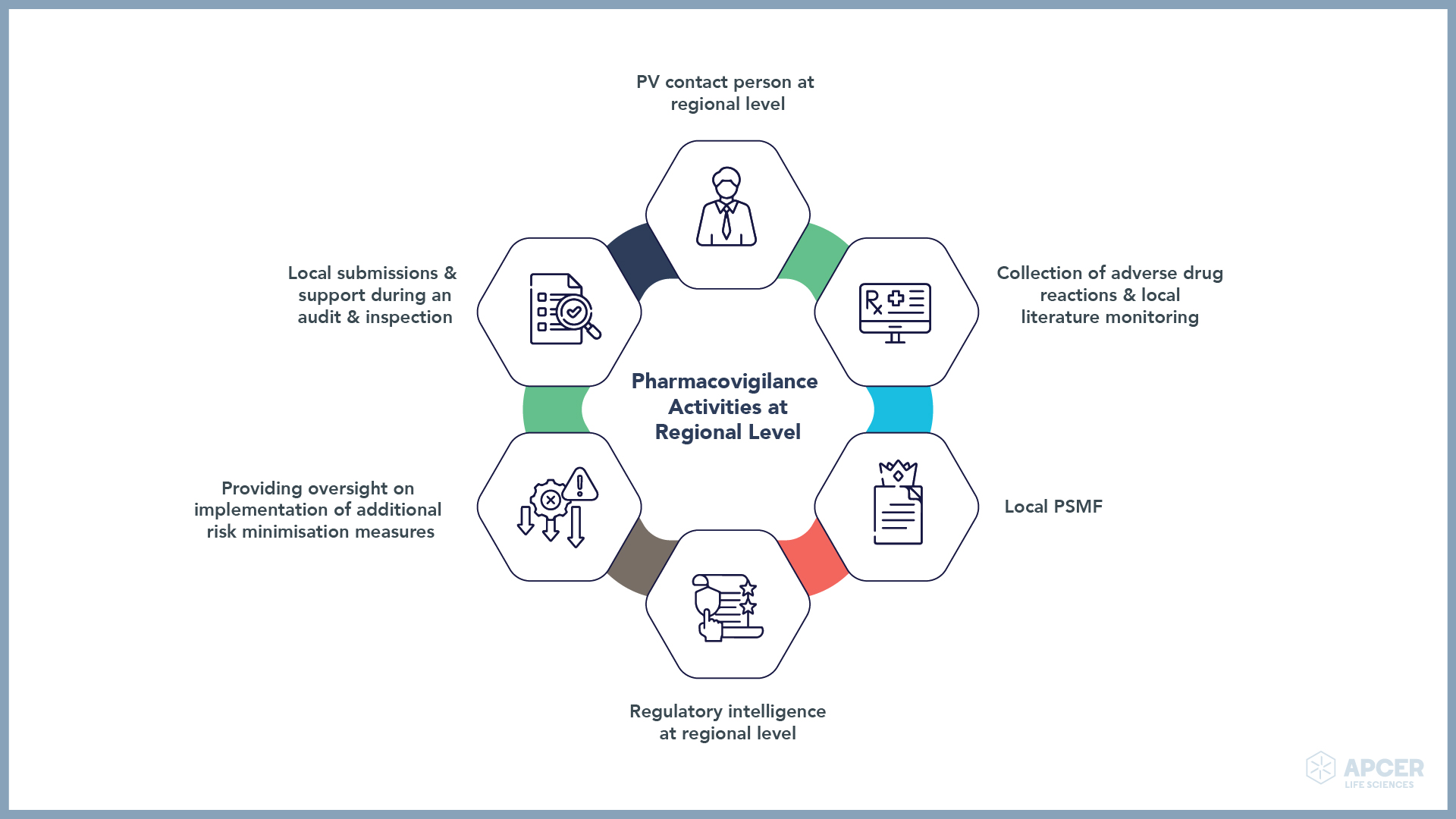
PV activities at regional level require the nomination of local QPPVs whose responsibilities include, but are not limited to, an oversight of day-to-day regional PV activities, collection of individual case study reports (ICSRs) and safety information from regional level, maintenance of local pharmacovigilance system master file (PSMF), and regular screening of local regulatory websites for any updates in legislations or guidance documents.
If multiple independent QPPV roles are involved in risk identification without a process or system in place, it may result in inconsistent and/or inefficient decision-making, as well as a lack of appropriate oversight. Further, the requirement for having an effective quality management system (QMS) in place becomes challenging by the worldwide oversight of numerous functions, which can easily lead to redundancy.
Also important is to integrate information on processes at local level into the global PSMF/EU PSMF. Collecting information from regional level and then collating it into global PSMF is also an ambitious task. Because the PSMF serves as a key tool for an effective oversight for the MAH and QPPV, integrating information from regional level into global PSMF becomes an essential step in PSMF management.
The ultimate goal is to perform all PV activities at regional level as a part of global PV system maintenance and global QPPV oversight. PV activities performed at regional level have a direct impact on patient safety.
Solutions
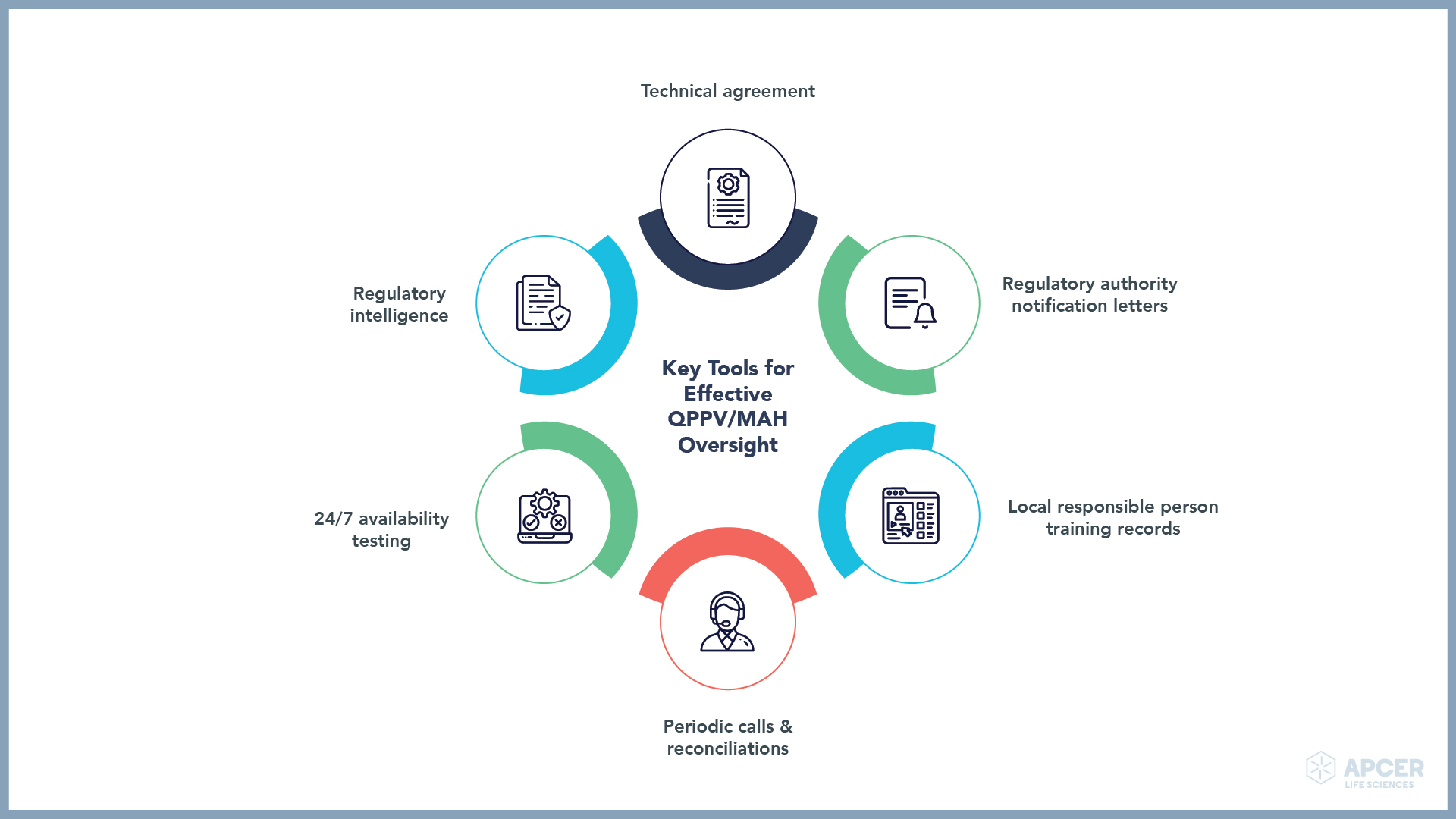
It is the responsibility of the MAH and QPPV to nominate a PV contact person at regional level/a local QPPV. This local QPPV should have a functional reporting to the EU QPPV/global QPPV for medicinal products authorised with respect to PV activities being performed at regional level. The infographic below shows the key tools or processes that can help in effective oversight.
PV documents, such as technical agreements, can be executed, which describe roles and responsibilities of each party involved. Regulatory intelligence is an important tool that helps in the identification of local legislation or local safety updates, which can be shared with global teams. Timely notification of changes in legislation helps in performing an impact analysis for current processes and incorporating changes into global and local procedures without any delay. Periodic calls and routine reconciliations help keep local and global PV teams connected continuously.
Local QPPVs/local responsible persons should be integrated as globally qualified strategic partners into the PV system. This helps in the maintenance of global PSMF, which fulfils various needs of the MAH and QPPV in keeping an oversight of the PV system.
The network of regional partners helps fostering expertise and local coverage for effective maintenance of the PV system.