Pragmatic Approaches
to Risk Minimization Measures
Every therapeutic modality has associated risks. These risks, if not managed well, can pose threat to patient safety or even public health and regulators take this very seriously.
Regulators have mandated proactive risk assessments and continuous safety surveillance for pharmaceutical drugs throughout their life cycle. Keeping the risk management plan (RMP) up to date is vital; however, a mere update does not solve the purpose unless RMP commitments, especially risk minimization measures (RMM) are designed, implemented, and utilized well.
An efficient risk minimization system lays out a systematic platform on which the pharmacovigilance plan and risk–benefit analysis are characterized for any drug.
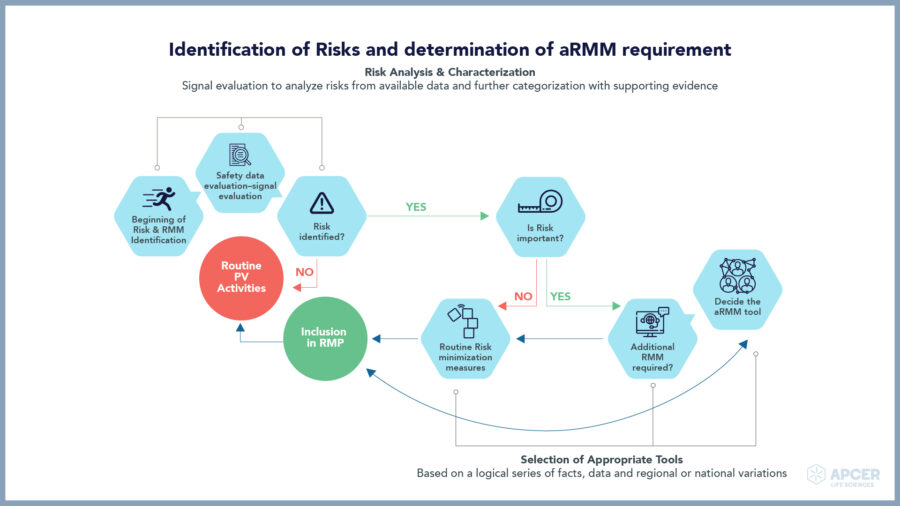
Identification of risks and determination of additional risk minimization measures requirement
Additional risk minimization measures (aRMMs) for medicinal products are necessary to address important safety issues, which may not be practically achieved through routine RMM (e.g., product information, pack size, or legal status) alone.
aRMMs are of following types:
- Educational materials
- Controlled access program
- Others (e.g., controlled distribution system, pregnancy prevention plan, etc.).
Such aRMMs are a resource-intensive activity; hence identification of right aRMMs and a practical implementation strategy would help in achieving the objective of risk minimisation.
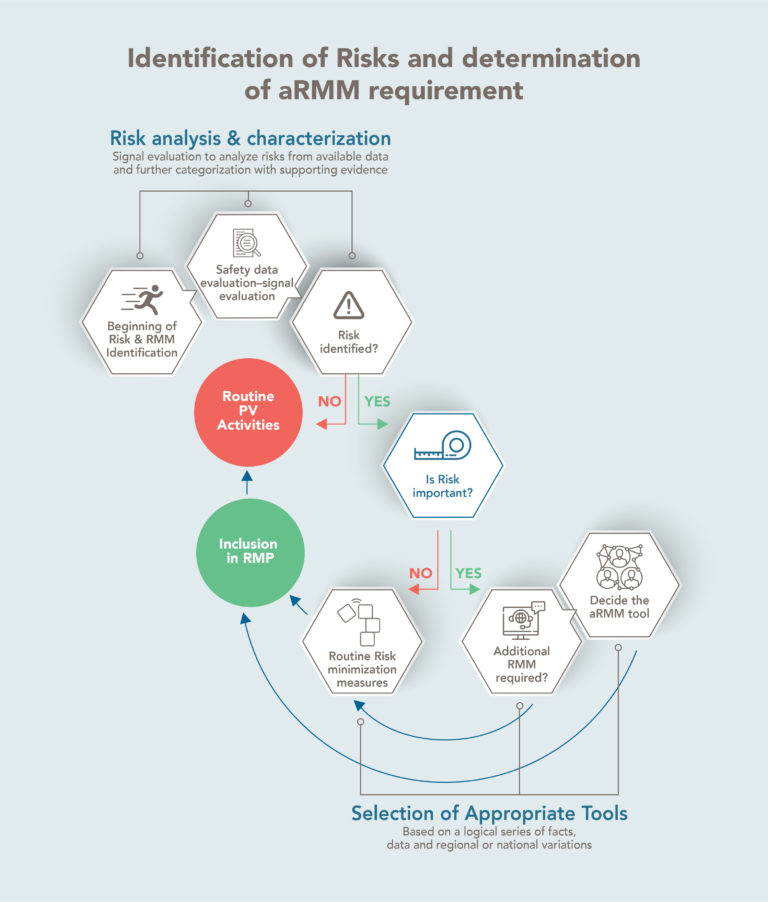
Post determination of aRMM requirements, leverage expertise of different stakeholders to create evidence-based dissemination and implementation of strategies into practice.
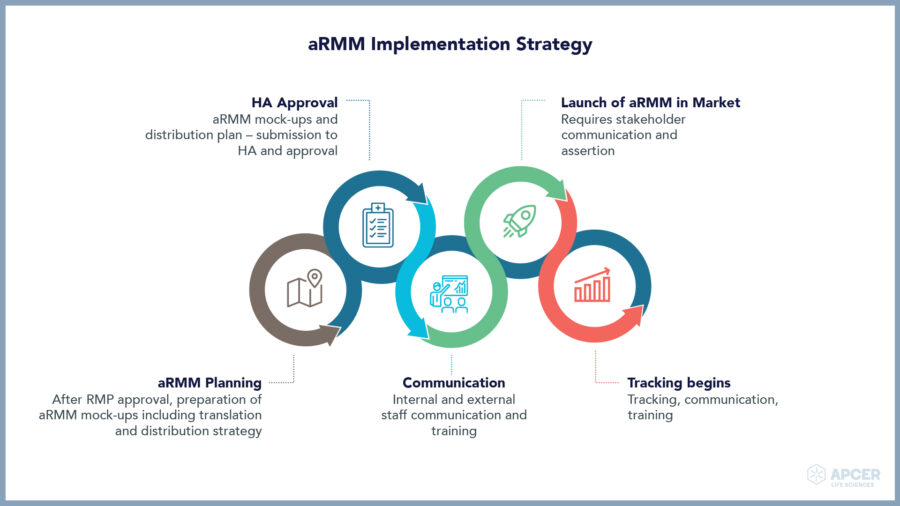
Implementation strategy of educational materials for medicinal products
Implementation and determination of the effectiveness of aRMMs can be a challenge as it involves multiple stakeholders. Once an aRMM tool is defined, a different course of journey begins as per selected aRMM tool. Upon analysis of the European Medicines Agency (EMA)-approved products in 2015 to 2017, 66% of the implemented aRMMs are of educational materials.(1)
Implementation strategies for educational materials incorporate a cascade of five events, as depicted below:
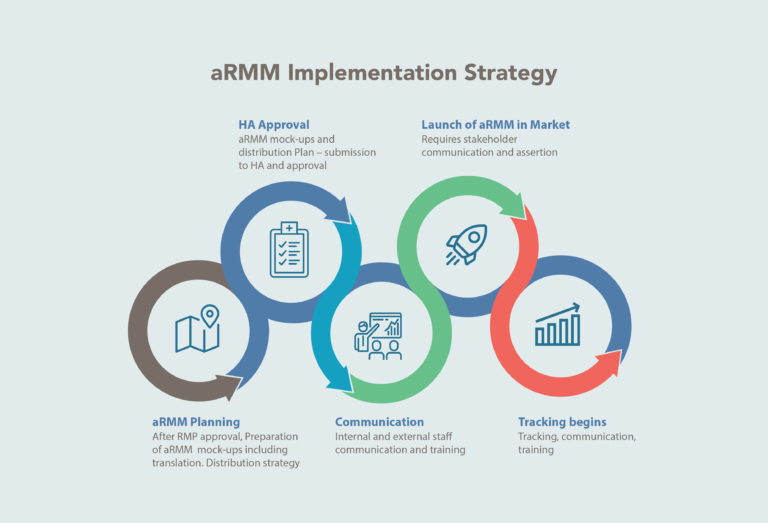
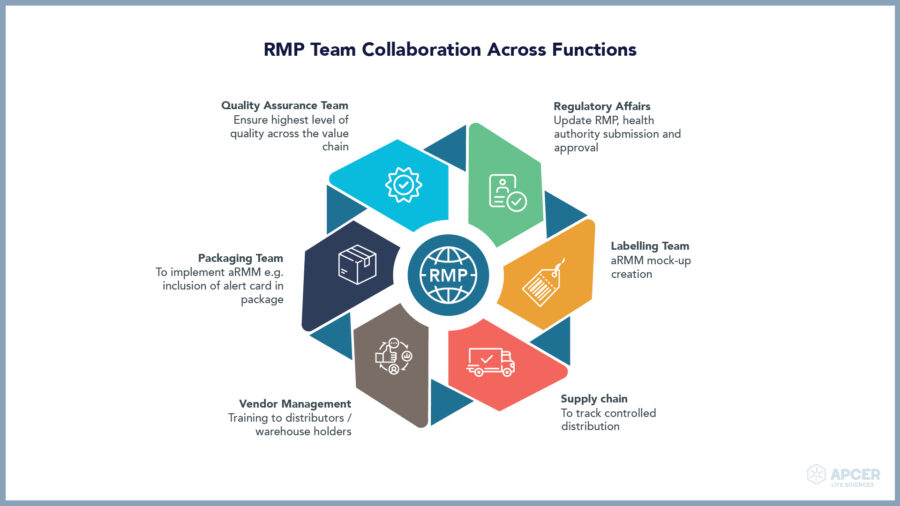
Safety stakeholders across functions
Risk management plan is an organization-wide document because safety is everyone’s responsibility. No matter how efficacious a product is, safety must be monitored. Hence, managing risks in a better way with the help of all stakeholders is a necessity. Success of any RMP commitments to adapt the interventions in routine practice depends upon collaboration and coordination among cross-functional groups. Execution of the central policy is necessary in the is necessary for the biopharmaceutical companies to keep global and local teams in sync.
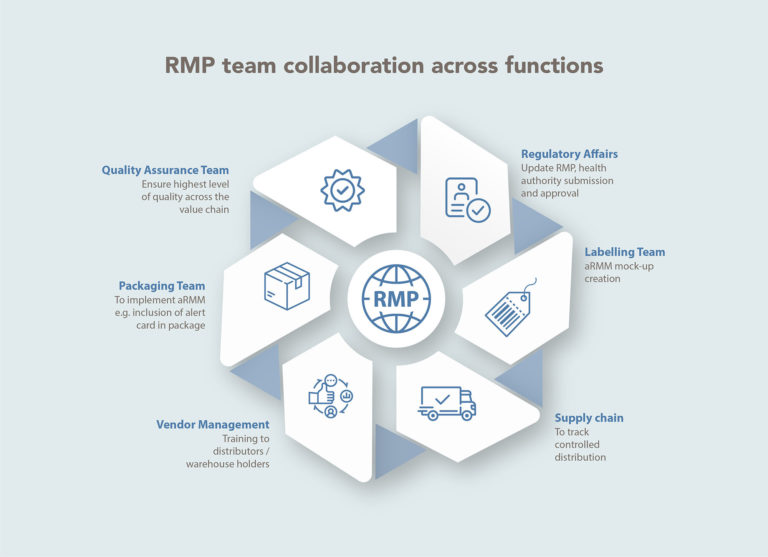
APCER Life Sciences – Your preferred partner for Risk Management
- Dedicated team of healthcare professionals, including physicians, for risk assessment and evaluation.
- 12+ years of rich experience in strategizing, designing and implementing global risk management systems.
- Deep knowledge of specialized services such as aRMM tracking, effectiveness assessment, supports consortium, Targeted Follow up Questionnaire (TFUQs), periodic review, etc.
- Marketing authorization holder (MAH)/Applicant support for oversight process build-up, stakeholder/local affiliate training, implementation hand holding, and risk group committee workshop
- Collaborative service model covering end-to-end services and specialized support, e.g., subject matter expert (SME) review or gap analysis.
To know more on how we can help you implement an effective risk management system, connect with our experts today.