FAQs about
Frequently Asked Questions about In-vitro Diagnostic Medical Devices (IVD) and In vitro diagnostic medical device regulation (IVDR)
1. What is IVD?

2. What is CE marking?
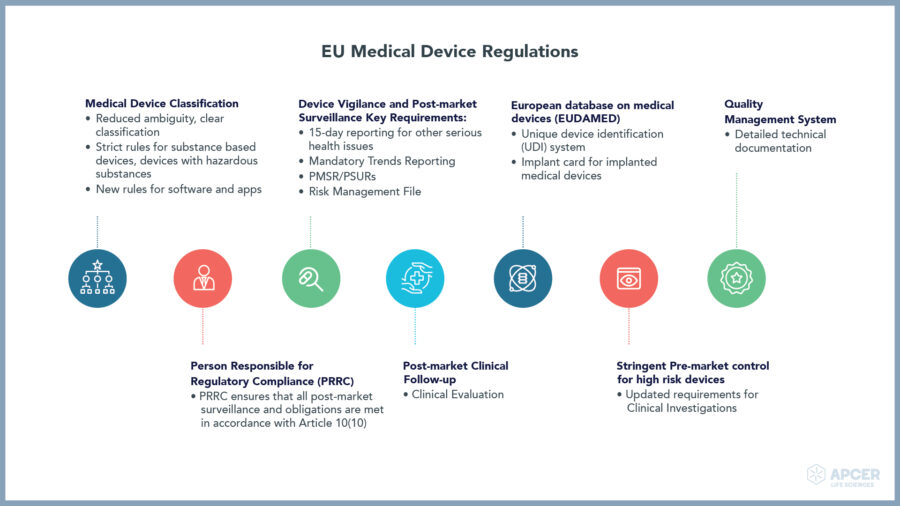
In the European Union (EU), medical devices must undergo a conformity assessment for meeting legal requirements to ensure that they are safe to use and exhibit intended performance. These are regulated at the level of EU member state who designates accredited notified bodies to conduct conformity assessment; however, they should seek European Medicines Agency (EMA) scientific opinion before issuing a CE (Conformité Européenne) certificate.
Once a medical device has passed a conformity assessment, the manufacturers can place CE mark on that medical device.
3. What is UDI system?
Unique Device Identification System assigns a unique device identifier (UDI) for every IVD device; this allows the identification and facilitates the traceability of devices.
4. What is the main reason for IVDR implementation?
In vitro diagnostic medical device regulation (IVDR) creates a robust, transparent, and sustainable regulatory framework that is recognized internationally, improves clinical safety, and creates fair market access for manufacturers and healthcare professionals.
5. How are devices classified under IVDR?

6. When will medical device manufacturers need to comply with the new IVDR?
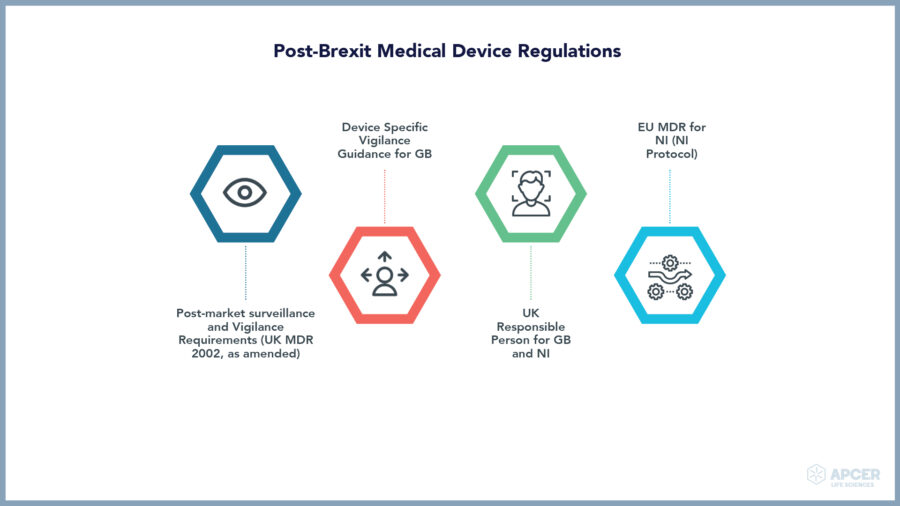
IVDR (EU) 2017/746 will apply from 26 May 2022.
7. What are the conditions to benefit from the extended transition period for IVDR?
IVDR provides transitional period until 26 May 2024 for IVDs with a certificate issued by a notified body in accordance with the directive on IVDs prior to 26 May 2022.
8. What is a notified body?
A notified body is an organization designated by an EU country to assess the conformity of certain products before bringing them in the market. These bodies carry out tasks related to conformity assessment procedures set out in the applicable legislation when a third party is required.
References:
https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX:02017R0746-20170505. Last accessed on 15 Apr 2022,
https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX:02017R0745-20170505. Last accessed on 15 Apr 2022.
Published on: April 28, 2022
Explore More
Testimonials

I must commend the organizational skill, the professionalism, the clarity, the expertise on subject matter, execution excellence that APCER has exhibited in all my projects where I had collaborated…
Medical Advisor, Medical Affairs
A leading bio-pharma company
My experience with APCER has been excellent. The team was very helpful, knowledgeable, responsive and professional with all the work that was needed.
Global Clinical Leader
A leading pharmaceutical company
APCER demonstrated extreme flexibility accommodating changes, and a number of last-minute changes were implemented quickly and correctly. I also appreciate the flexible communication during my workday, given the time zone differences between the APCER writers and myself.
Associate Submission Medical Writer
A leading bio-pharma company
APCER’s speed, responsiveness and flexibility to meet our safety needs is something we have always valued. APCER has helped harmonize our global pharmacovigilance operations and made us compliant with ever changing regulations.
Medical Director
US-based specialty pharmaceutical company
The audit was carried out professionally and objectively. The scope of the audit was relevant and appropriate, and time efficient. The discussions during the audit were constructive and recommendations where helpful and appropriate. The team members involved in the audit had a positive experience and were made to feel at ease by the auditors.
QPPV and Pharmacovigilance Head

We are thankful to team APCER for bringing in strong practices & helping inspection readiness in our pharmacovigilance framework which led to successful FDA audits.
Senior Director
US-based pharmaceutical company
APCER’s invaluable support with Clinical trial disclosure deadlines helped us in timely submissions of periodic reports.
Head, Clinical Trial Transparency
Top 10 pharmaceutical company with operations in the EU and US
We appreciate APCER for its efficiency in responding to matters that they get even at the eleventh hour. Working with APCER has made compliance a lot easier.
AVP, Pharmacovigilance
US-based pharmaceutical company
We are thankful to team APCER for bringing in strong practices & helping inspection readiness in our pharmacovigilance framework which led to successful FDA audits.
Senior Director
US-based pharmaceutical company