Background
Regulatory agencies have been working closely with pharmaceutical companies to support and expedite approval of innovative products and therapies. They are increasingly focusing on strengthening the approach and methods for the assessment of quality, safety, and efficacy of drugs, devices, and combination products. To meet unmet needs for novel, safe drugs and therapies, drug safety and regulatory departments at pharmaceutical companies work together to navigate and operate in a dynamic but continuously stringent regulatory environment.
Meeting complex regulatory requirements for drug safety can be challenging for all sizes of organizations. In addition to this, there are other challenges including financial constraints and pressures to advance products through discovery and clinical development, submission of applications to regulatory agencies, and developing successful launch plans to bring safe and effective products to patients. Successfully managing the drug safety profile of your program throughout the drug lifecycle is an asset. Having access to aggregate information across studies and at the same time reducing overhead and redundant fees pays dividends each time regulatory reports, scientific communications, and applications are prepared. Due diligence for partnership and investment purposes can be packaged for accurate financial assessment. Executives discuss knowns and unknowns with the assurance that a complete safety analysis of the data has been performed. Therefore, it is important to partner with a specialized pharmacovigilance services provider to review and analyze consolidated data, help to define the initial product label, and proactively identify, assess, and help to manage safety concerns.
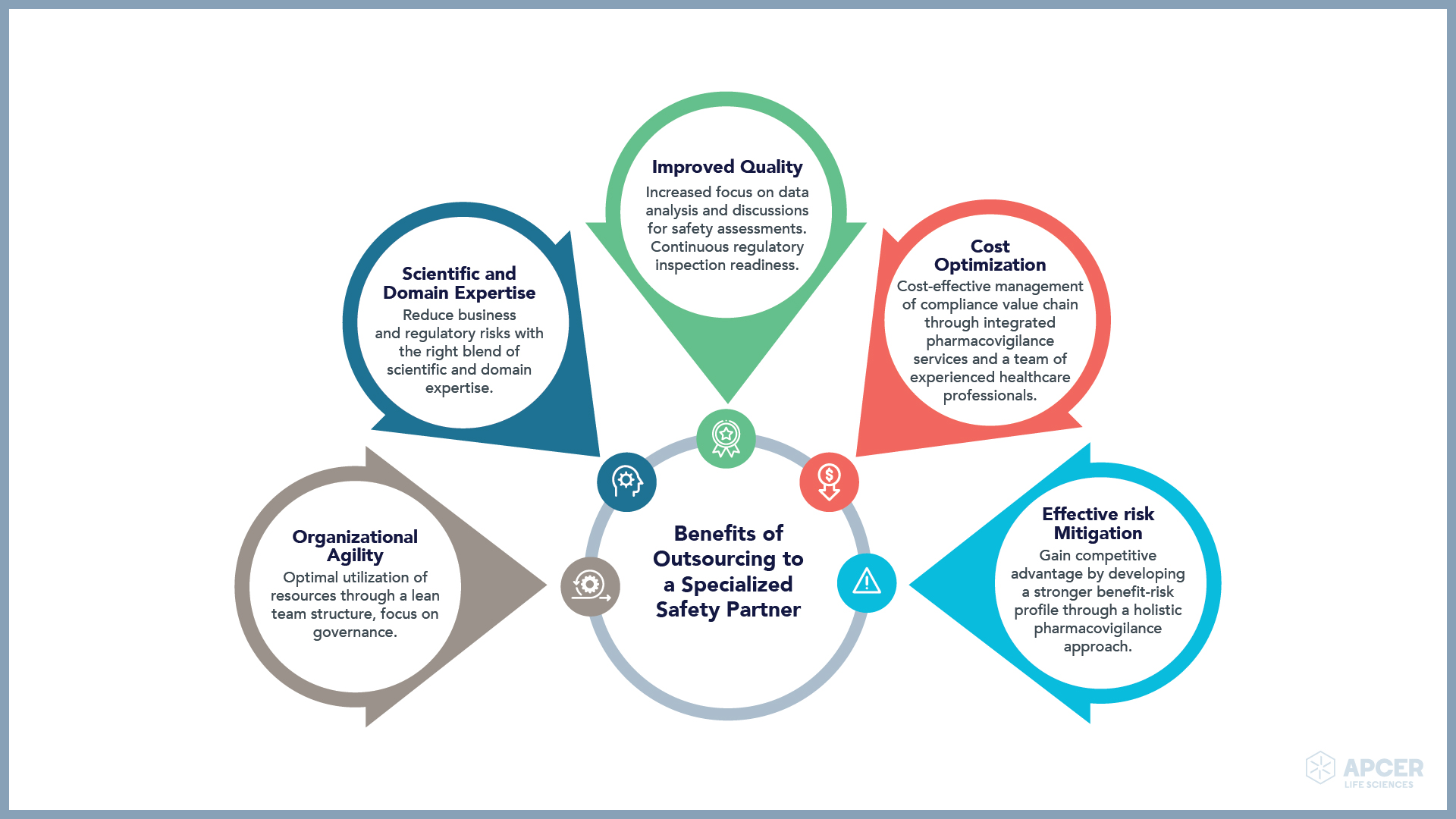
Partnering with and outsourcing a critical function such as drug safety to a specialized pharmacovigilance services provider can help pharmaceutical companies in the following ways:

Organizational Agility
As pharmaceutical and biotechnology companies introduce new products into clinics and subsequently the commercial market(s), their requirements for pharmacovigilance services evolve. An experienced and expert pharmacovigilance services provider enables a lean team structure, focuses on governance, and leverages the team’s scientific and medical expertise. This helps the innovator company to optimally utilize their resources.

Scientific and Domain Expertise
There is a growing emphasis on overall quality, accurate data collection and reporting of safety information. It becomes imperative for biopharma companies to provide high quality case processing and reporting. This challenge is amplified with handling of large case volumes and meet signalling, surveillance, and risk management requirements. A specialized safety partner can offer experienced and skilled resources that can help create a seamless and efficient pharmacovigilance program. Right blend of scientific and domain expertise can help reduce business and regulatory risk by accelerating the drug approval process and time-to-market.

Improved Quality
In an ever-changing regulatory environment, biopharma companies are expected to maintain quality, compliance, and inspection readiness of their processes. An experienced pharmacovigilance services provider can play a crucial role in enabling companies to adopt the best industry practices and strengthen processes. The service partner can help generate high-quality narratives and maintain clinical source data which enable sponsors to aggregate data effectively. This will as a result help to focus on data analysis and discussions for safety assessments and continuous regulatory inspection readiness.

Cost Optimization
In order to maintain compliance, companies need to recruit an experienced and skilled workforce with ongoing knowledge of varied regulatory obligations and global reporting requirements. Experienced professionals are challenging to hire and retain. A range of experience and expertise may be needed at different stages of drug development, asset acquisition, or post marketing.
A pharmacovigilance services provider who behaves as a partner offers integrated services and an ongoing team consisting of trained and experienced healthcare professionals who manage the compliance value chain in a cost-effective manner. They can also help provide a customized model for hosting end-to-end pharmacovigilance (PV) safety database, sustainable ramp-up of services, and fast-track implementation of regulatory intelligence. They bring industry best practices and global experience and deepen the discussion around the safety of the product.

Effective Risk Mitigation
The specialized safety partner offers robust processes that take a holistic approach towards the pharmacovigilance quality system. Such a system would support audits as well as risk assessments and mitigation strategies.
As newer drugs and therapies are developed, tested, and approved, biopharmaceutical companies will be required to meet stringent requirements for patient safety. A specialized pharmacovigilance services partner at the early stages of drug development throughout the post-marketing cycle, enables companies to remain compliant, better understand the safety profile of their product, and react to provide safe and effective therapies to patients. Biopharma companies may even gain a competitive advantage by developing a stronger benefit–risk profile and improved identification of at-risk patients.